Surfactants are key cleaning agents that break down grease and dirt using their unique molecular structure. They have a hydrophilic head that attracts water and a hydrophobic tail that binds to oils and fats. This allows them to form micelles, trapping grime so it can be rinsed away easily. Understanding how surfactants work helps you choose safer, more effective products. Keep exploring to learn more about how these molecules really clean your surfaces and skin.
Key Takeaways
- Surfactants contain hydrophilic heads and hydrophobic tails that form micelles to trap and remove grease and oils.
- They reduce surface tension, allowing water to penetrate and loosen stubborn dirt and greasy residues.
- Surfactants surround lipids and oils within micelles, facilitating encapsulation and easier rinsing away.
- Different types, like anionic and nonionic, offer varied degreasing effectiveness and gentleness on surfaces.
- Biodegradable surfactants provide effective cleaning while minimizing environmental impact through eco-friendly formulations.
What Are Surfactants and Why Are They Key to Cleaning?

Surfactants are the essential ingredients that make cleaning effective because they help break down and remove dirt, grease, and oils from surfaces. When choosing surfactants, biodegradability considerations are vital, as they impact environmental impact and sustainability. Eco-friendly surfactants break down more easily in nature, reducing pollution. During surfactant manufacturing, companies focus on creating formulas that balance cleaning power with environmental responsibility. This involves selecting raw materials that are biodegradable and minimizing harmful chemicals. Your choice of surfactants can influence not only cleaning efficiency but also ecological health. Understanding the environmental impact of surfactants and their production processes can help consumers and manufacturers promote more sustainable practices. By understanding how surfactants are produced and their environmental impact, you can make more informed decisions about the cleaning products you use. This knowledge supports sustainable practices while ensuring your surfaces stay spotless.
How Do Surfactants Dissolve Grease and Dirt?

When surfactants encounter grease or dirt, they form tiny clusters called micelles that trap the grime. These micelles surround oily lipids and oils, breaking them into smaller, more manageable pieces. This process makes it easier for water to wash away the dirt and grease effectively. Effective cleaning relies on the surfactants’ ability to emulsify and suspend contaminants, ensuring a thorough clean. Additionally, understanding the chemistry of surfactants helps in designing more efficient cleaning products. Recognizing the durability and functionality of surfactant formulations is crucial for optimizing cleaning performance across various applications. Proper formulation also considers filter maintenance to prevent buildup and maintain effectiveness over time. Moreover, selecting energy-efficient appliances can enhance overall cleaning efficiency and reduce power consumption during household chores.
Micelle Formation Process
To understand how surfactants dissolve grease and dirt, it helps to know what happens at the microscopic level. When surfactants are added to water, their molecules undergo molecular dynamics, where the hydrophobic tails seek out oils and grease, while the hydrophilic heads stay in water. This process forms micelles—tiny spherical structures that trap dirt and grease inside. During surfactant synthesis, molecules are designed to optimize this process, enhancing cleaning power. The formation of micelles is driven by the balance between attractive and repulsive forces, making dirt soluble in water. Here’s a quick overview:
| Step | Description | Result |
|---|---|---|
| Surfactant molecules align | Hydrophobic tails inward, hydrophilic heads outward | Micelle formation begins |
| Micelle assembly | Structures trap oils and dirt | Dirt becomes water-soluble |
| Stabilization | Micelles remain suspended in water | Effective cleaning |
| Detachment | Dirt-laden micelles are rinsed away | Clean surface |
Additionally, surface tension reduction plays a crucial role in allowing surfactants to penetrate and loosen stubborn grime effectively. Understanding micelle formation is essential for appreciating how surfactants achieve their powerful cleaning action.
Lipid and Oil Interaction
Ever wonder how grease and oil dissolve so effectively in water? It all comes down to lipid interactions and oil compatibility. Surfactants have unique molecules with hydrophobic tails that attract oils and lipids, breaking them apart from surfaces. These tails interact with grease and dirt’s lipid structure, effectively dissolving them into tiny droplets. Because of their affinity for oils, surfactants form stable emulsions, preventing the grease from re-adhering to surfaces. This process relies on the surfactant’s ability to bridge the gap between water and oily substances, making the lipids more accessible to water molecules. Essentially, surfactants manipulate lipid interactions and leverage oil compatibility, transforming stubborn grease into manageable particles that water can wash away easily. Additionally, the effectiveness of this process can depend on factors such as molecular structure, concentration, and surface tension properties of the surfactants used. The emulsification process is crucial in maintaining the stability of these oil-in-water mixtures, ensuring thorough cleaning. Furthermore, the interfacial tension between oil and water plays a significant role in how efficiently surfactants can break down greasy residues. The surface chemistry involved influences the stability and breakdown of greasy substances during cleaning.
What Is the Structure of Surfactants? Hydrophilic Heads and Hydrophobic Tails
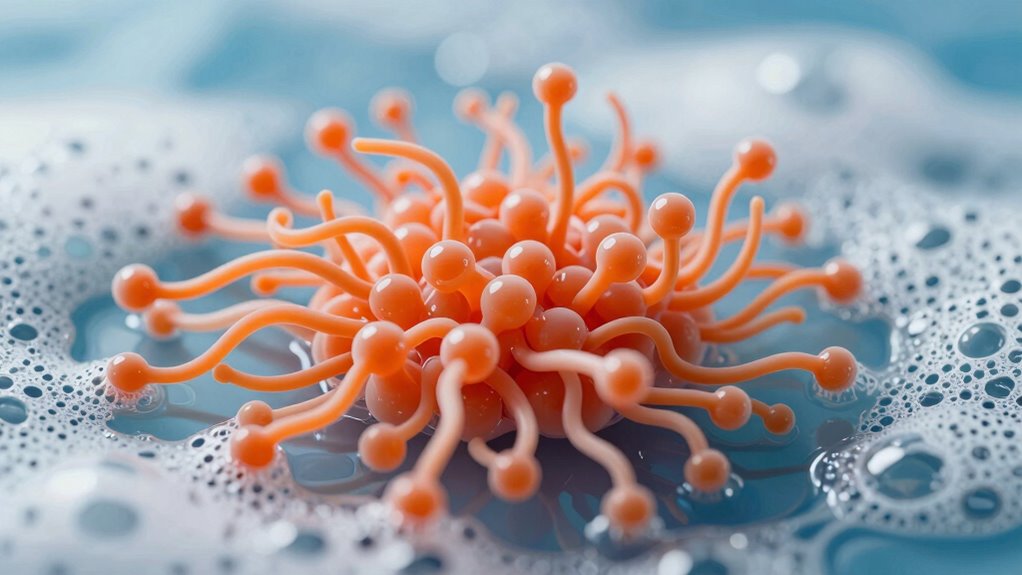
Surfactants are composed of two distinct parts: a hydrophilic head and a hydrophobic tail. Your focus should be on their molecular arrangement, which determines how they interact with water and oils. The hydrophilic head is attracted to water because of its polarity, making it water-loving. In contrast, the hydrophobic tail repels water and prefers to associate with oily or greasy substances. This polarity difference is key to how surfactants work—allowing them to bridge water and oil molecules, breaking down greasy dirt. The molecular arrangement enables surfactants to form structures like micelles, where the tails hide inside away from water, and the heads face outward. This structure is fundamental to their ability to clean effectively by lifting and suspending oils.
Types of Surfactants in Household Cleaners

You’ll find anionic surfactants in many household cleaners because they effectively remove dirt and grease. Nonionic surfactants are gentle yet versatile, making them great for sensitive surfaces and delicate fabrics. Understanding these types helps you choose the right cleaner for your specific needs.
Anionic Surfactants Roles
Anionic surfactants play a crucial role in household cleaners by effectively breaking down and removing dirt, grease, and stains. Their negative charge helps attract and lift oily substances from surfaces. When choosing products, consider biodegradability concerns, as some surfactants may persist in the environment. The synthesis process affects both performance and ecological impact. Here’s a quick comparison:
| Surfactant Type | Key Feature | Environmental Impact |
|---|---|---|
| Sulfates | Good cleaning power, foaming | Moderate biodegradability |
| Sulfonates | Strong stain removal, low cost | Varies by synthesis method |
| Alkyl Benzene Sulfonates | Widely used, economical | Biodegradability varies |
| Linear Alkylbenzene Sulfonates | Better biodegradability | Improved in newer formulations |
| Alcohol Ether Sulfates | Mild, suitable for sensitive skin | Generally biodegradable |
Choosing the right anionic surfactant balances cleaning efficacy with environmental considerations. Water-based environments can influence the effectiveness and environmental impact of these surfactants.
Nonionic Surfactants Benefits
Nonionic surfactants are widely favored in household cleaners because they effectively remove dirt and grime without generating excessive foam or causing skin irritation. They work well across various temperatures and pH levels, making them versatile. One significant benefit is their lower environmental impact, as many nonionic surfactants are biodegradable, breaking down more easily in nature. This biodegradability reduces pollution and supports eco-friendly cleaning practices. Additionally, they tend to be less harsh on skin and surfaces, making them safer for everyday use. Their gentle yet effective cleaning power, combined with better environmental impact, makes nonionic surfactants a popular choice in eco-conscious households. Overall, they balance cleaning performance with sustainability, helping you maintain a clean home responsibly.
How Do Surfactants Lower Surface Tension for Better Cleaning?

Ever wonder how surfactants make cleaning so much easier? It all comes down to reducing surface tension. Surface tension is the force that holds molecules together at a liquid’s surface, making water bead up instead of spreading out. Surfactants lower this tension by positioning their molecules at the water’s surface, with their hydrophobic tails sticking out and their hydrophilic heads remaining in the water. This action breaks up the cohesive forces, allowing water to spread more easily and penetrate dirt and grime. By decreasing surface tension, surfactants improve detergent efficiency, helping water reach into tiny crevices and lift away stubborn dirt. This process makes cleaning faster, more effective, and less labor-intensive.
How Do Surfactants Help Remove Oil and Grease?

Surfactants form tiny clusters called micelles that trap oil and grease molecules inside. These micelles encapsulate the stubborn oils, making them easier to wash away. This process helps clean surfaces by breaking down and removing greasy residues efficiently. Additionally, understanding the science behind surfactants highlights their crucial role in effective cleaning. Their effectiveness is also supported by attention to detail in formulation and application, ensuring thorough removal of contaminants.
Micelle Formation Mechanism
When surfactants encounter oil and grease, they spontaneously organize into structures called micelles, which are vital for cleaning. These micelles are formed because surfactants are cleansing agents with both hydrophilic heads and hydrophobic tails. The hydrophobic tails are attracted to the oil and grease, while the hydrophilic heads face outward toward the water. This arrangement reduces the surface tension between oil and water, allowing the oil to disperse into tiny droplets within the micelle. As a result, the grease becomes trapped inside these spheres, making it easier to wash away. Micelle formation is crucial because it transforms stubborn oils into manageable particles, enabling water and surfactants to lift and rinse away grease efficiently. Additionally, the stability of micelles depends on factors like surfactant concentration and temperature, which can influence cleaning effectiveness. Consistent practice of using surfactants can improve cleaning efficiency over time. Moreover, understanding surfactant properties can help optimize cleaning processes for different types of oils and greases. Recognizing the role of molecular structure can further assist in selecting the most effective cleaning agents for specific tasks.
Oil and Grease Encapsulation
Surfactants play a crucial role in removing oil and grease by encapsulating these stubborn substances within micelles. When you apply a surfactant, its molecules surround the oil or grease, forming tiny micelles that trap the filmy residues. This process makes it easier to rinse away dirt, leaving surfaces clean. Eco-friendly alternatives are increasingly available, reducing environmental impact without sacrificing effectiveness. Advances in surfactant synthesis focus on creating biodegradable and sustainable options that perform well in degreasing tasks. By understanding how surfactants encapsulate oil and grease, you can choose formulations that are both efficient and environmentally responsible. This encapsulation process is the key to effective degreasing, making surfactants essential in household cleaning, industrial applications, and eco-conscious products.
Are Surfactants Safe for Skin and the Environment?

Many people wonder whether surfactants are safe for their skin and the environment. Concerns about biodegradability and skin irritation potential often come up. First, some surfactants may not break down easily, raising biodegradability concerns that can harm aquatic ecosystems. Proper formulation and ingredient selection are crucial in ensuring surfactants are both effective and safe. Second, certain formulations can cause skin irritation, especially for sensitive skin types. Third, advances in surfactant chemistry aim to reduce these risks by creating more environmentally friendly and gentle options. By choosing surfactants that are biodegradable and mild, you can minimize environmental impact and skin sensitivities. Biodegradability is an essential factor to consider when selecting surfactants, as it directly affects their impact on the environment. Always check product labels for eco-friendly certifications and ingredient lists to ensure you’re making safe choices. Understanding biodegradable surfactants can help you make more informed decisions that benefit both your skin and the planet. Incorporating eco-friendly ingredients can further enhance the safety and sustainability of your cleaning products. With informed selection, surfactants can be both effective and safe for you and the planet.
How to Choose Safe and Effective Surfactant-Based Products

Choosing safe and effective surfactant-based products starts with reading labels carefully. Look for information on biodegradability concerns to guarantee the product breaks down naturally and minimizes environmental impact. Also, consider surfactant sourcing; ethically sourced ingredients reduce ecological harm and support sustainable practices. To help evaluate options, review this quick guide:
| Aspect | What to Check |
|---|---|
| Biodegradability | Look for labels indicating biodegradable ingredients. |
| Surf. Sourcing | Choose products with transparent sourcing info. |
| Effectiveness | Select formulas suited for your cleaning needs. |
Being vigilant about these factors ensures you pick products that are safe for you and the environment while maintaining cleaning power.
Myths and Facts About Surfactants in Cleaning Products

While reading labels helps you select safer, more sustainable cleaning products, there are still common misconceptions about surfactants that can lead to confusion. One myth is that all surfactants are harmful to the environment; in reality, many are biodegradable and pose minimal environmental impact when used properly. Second, some believe surfactants compromise consumer safety; however, most are tested rigorously to ensure they’re safe for household use. Finally, people often think natural surfactants are less effective; in fact, many plant-based options perform just as well as synthetic ones. Understanding these facts helps you make informed choices, balancing environmental impact and consumer safety. Additionally, awareness of Global Chaos – Microsoft Outage incidents emphasizes the importance of reliable products and contingency planning in everyday life. Clarifying these misconceptions allows you to choose cleaning products that are both effective and environmentally responsible.
What’s Next? Innovations in Eco-Friendly Surfactants

As researchers and companies seek sustainable solutions, innovation in eco-friendly surfactants accelerates, leading to new formulations that are both effective and gentle on the environment. Biodegradability advancements play a vital role, ensuring surfactants break down quickly without harming ecosystems. Plant-based formulations are at the forefront, replacing traditional petrochemicals with renewable, naturally derived ingredients. These innovations not only reduce environmental impact but also meet consumer demand for safer, greener cleaning products. By harnessing sustainable raw materials and improving biodegradability, industry leaders are creating surfactants that deliver powerful cleaning while supporting ecological health. Expect to see more biodegradable, plant-based surfactants in the future, transforming the way we approach cleaning and environmental responsibility.
Frequently Asked Questions
How Do Surfactants Work at the Molecular Level During Cleaning?
You can understand how surfactants work during cleaning through molecular micelle formation and surfactant adsorption dynamics. Surfactants adsorb at the oil-water interface, reducing surface tension, and then form micelles that encapsulate grease and dirt. This process enables the molecules to lift away contaminants efficiently, making cleaning easier. The dynamics of adsorption and micelle formation are essential for breaking down stubborn grease, leaving surfaces clean and spotless.
Are All Surfactants Biodegradable and Environmentally Friendly?
Imagine a wave washing over a shoreline—some surfactants break down gracefully, leaving no trace, while others linger like stubborn debris. Not all surfactants are biodegradable or eco-friendly; some pose environmental concerns. You can choose eco-friendly alternatives, crafted to vanish effortlessly into nature’s embrace. By selecting these greener options, you help protect our planet, ensuring that cleaning doesn’t come at the expense of the environment’s delicate balance.
Can Surfactants Cause Skin Irritation or Allergies?
Yes, surfactants can cause skin irritation or allergies, especially if you have skin sensitivity concerns. You might experience unexpected allergic reactions or redness after using products with certain surfactants. To minimize risks, check labels for gentle or hypoallergenic options and avoid harsh formulations. If you notice irritation, discontinue use and consult a dermatologist. Being aware of your skin’s reactions helps protect you from potential adverse effects.
What Are the Differences Between Natural and Synthetic Surfactants?
Natural surfactants come from plant or animal sources, often making them more biodegradable and eco-friendly. Synthetic surfactants, on the other hand, are created through chemical synthesis, usually resulting in a faster production process but less biodegradability. You might find natural options gentler on skin, while synthetics can be more cost-effective and stable. Consider these factors when choosing based on biodegradability comparison and production methods.
How Do Surfactants Interact With Hard Water Minerals?
When you use surfactants in hard water, they interact with minerals through mineral binding, which can diminish their effectiveness. To improve cleaning, you often need water softening agents that remove or neutralize these minerals. This process prevents mineral buildup on surfaces and ensures the surfactants work efficiently, making your cleaning more effective and preventing soap scum or residue caused by mineral interactions.
Conclusion
Now that you understand how surfactants work like tiny knights battling grease, you can make smarter choices in your cleaning arsenal. Remember, not all surfactants are created equal—seek eco-friendly options to keep your home and planet safe. Just like the Great Library of Alexandria, knowledge is power. With this insight, you’re ready to conquer grime and make your cleaning routine more effective and responsible—no magic wand required!










